

Next: The function of
Up: Long Range Connections
Previous: Stimulus Dependent Synchronization:
In the simulations presented in the previous sections we have assumed
that the long range connections between cells with the same
orientation are excitatory. In reality, the nature of these long-range
connections is more complex, revealing both excitatory and inhibitory
influences [20]. For example, modulatory
influences on the response of a cell from outside its ``classical''
receptive field (CRF) have been observed in a number of physiological
experiments for both moving and stationary stimuli, both in cats and
primates [17,23,34]. In
most cases, presentation of extended arrays of similar stimuli leads
to a partial suppression of the cell response; response increases were
observed less frequently. Furthermore, the suppression is larger if
line segments outside the CRF have the same orientation as the
segments inside the CRF. A straightforward explanation of these
results in terms of a weak inhibitory influence of the
surround elements is, however, incompatible with other experimental
results [17,23] that show that
presentation of the same surround stimulus alone leads to a weak
excitation of the center cell.
We have recently addressed the balance between
excitation and inhibition in the long-range horizontal connections
[47] within the context of the network
model described above [47].
To simplify matters, we assumed
that the CRF maps onto a single hypercolumn in the V1 map of
orientation preferences, while surround stimuli excite
cells in neighboring hypercolumns. Given the different characteristics
of excitatory and inhibitory cells, the response to additional
input through the lateral connections depends on the firing
rate of the local population responding to the CRF stimulus.
More explicitly, for very weak
(sub-threshold) `center' stimulation , a surround stimulation of the same
orientation as the center stimulation is mainly
excitatory, while for a strong center stimulus the surround contributes
mainly inhibition (see Figure 13),
explaining thus the experimental data reported above.
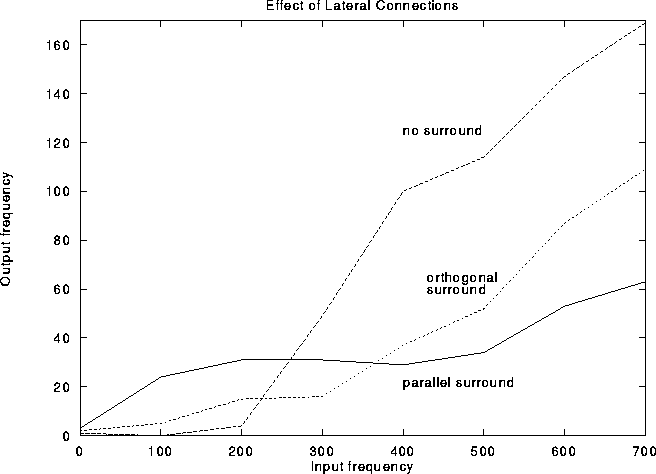
Figure 13: Average response curves (``F-I curves'') of cells within
the classical receptive field as a function of LGN
input frequency. The central stimulus is always at the
preferred orientation. The dashed curve is in the
absence of any additional input. The dotted and full lines were
obtained by adding orthogonal and parallel surround stimuli,
respectively. Note that all surround input enhances the
response frequency for low input and suppresses the response for large
center input. Note, furthermore, that surround input always suppresses
large inputs but more so for same-orientation surround input than for
orthogonal surround input.
As we further discuss in [47], this dual
effect is a result of the stochastic nature of the spiking input to
cells (stochastic resonance) and of the differential excitability of
pyramidal and stellate cells and inhibitory neurons.
The computational consequence of activity-dependent modulation is
obvious. An efficient information processing system should enhance
local differences by suppressing the response to homogeneous stimuli,
thereby reducing input redundancy. Such effects may correspond to
psychophysical observations of pop-out [5] and
tilt-illusion phenomena [59] and seem to require
inhibition between cells with similar orientation preferences. On the
other hand, when the system is presented with degraded information or
weak stimuli, the most efficient strategy consists in performing
correction processes such as line completion and stimulus enhancement.
Indeed, [40,41] showed that the detection
threshold of an oriented stimulus is lowered by the presence of
collinear ``flanking'' stimuli with the same orientation. Such an
effect is consistent with excitation between orientation columns tuned
to similar orientations (modulating a weak stimulus).
As shown in Figure 14, simulations show that the
excitatory surround contribution (obtained when the center-stimulation is weak)
indeed leads to a decrease in the detection threshold.
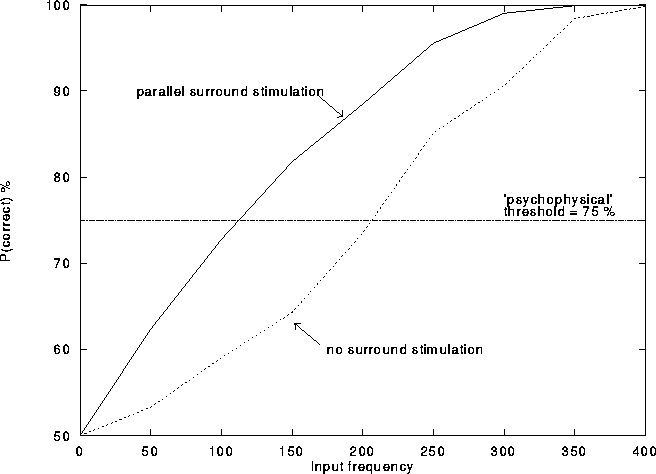
Figure 14:
The addition of parallel surround stimulation lowers
the detection threshold of the center stimulus at weak input intensities.
300 trials were run under each stimulus condition; the spike count
of one cell within 150 msec of stimulus onset was recorded for each
trial. For each surround stimulus condition, the curve represents
the probability, given any two trials, one with a stimulus,
and one without, that the trial with the stimulus produced
more spikes than the trial without a central stimulus.


Next: The function of
Up: Long Range Connections
Previous: Stimulus Dependent Synchronization:

